
Brownmed launches Body Furnace: A hot, new product in outdoor gear
Brownmed has introduced Body Furnace, the next generation, app-controlled personal heating device that goes wherever life takes the wearer.

Brownmed has introduced Body Furnace, the next generation, app-controlled personal heating device that goes wherever life takes the wearer.

Next week, Budweiser and Jim Beam Bourbon will recognize and celebrate a historic day in America – Repeal Day – the official end of Prohibition.

Unilever has announced that chief executive officer Paul Polman has decided to retire from the company.

The demand for CBD-based products is steadily rising as the cannabis market continues to gain momentum via sweeping legalization in North America, as well as natural economic growth as it settles into the mainstream consumer markets.
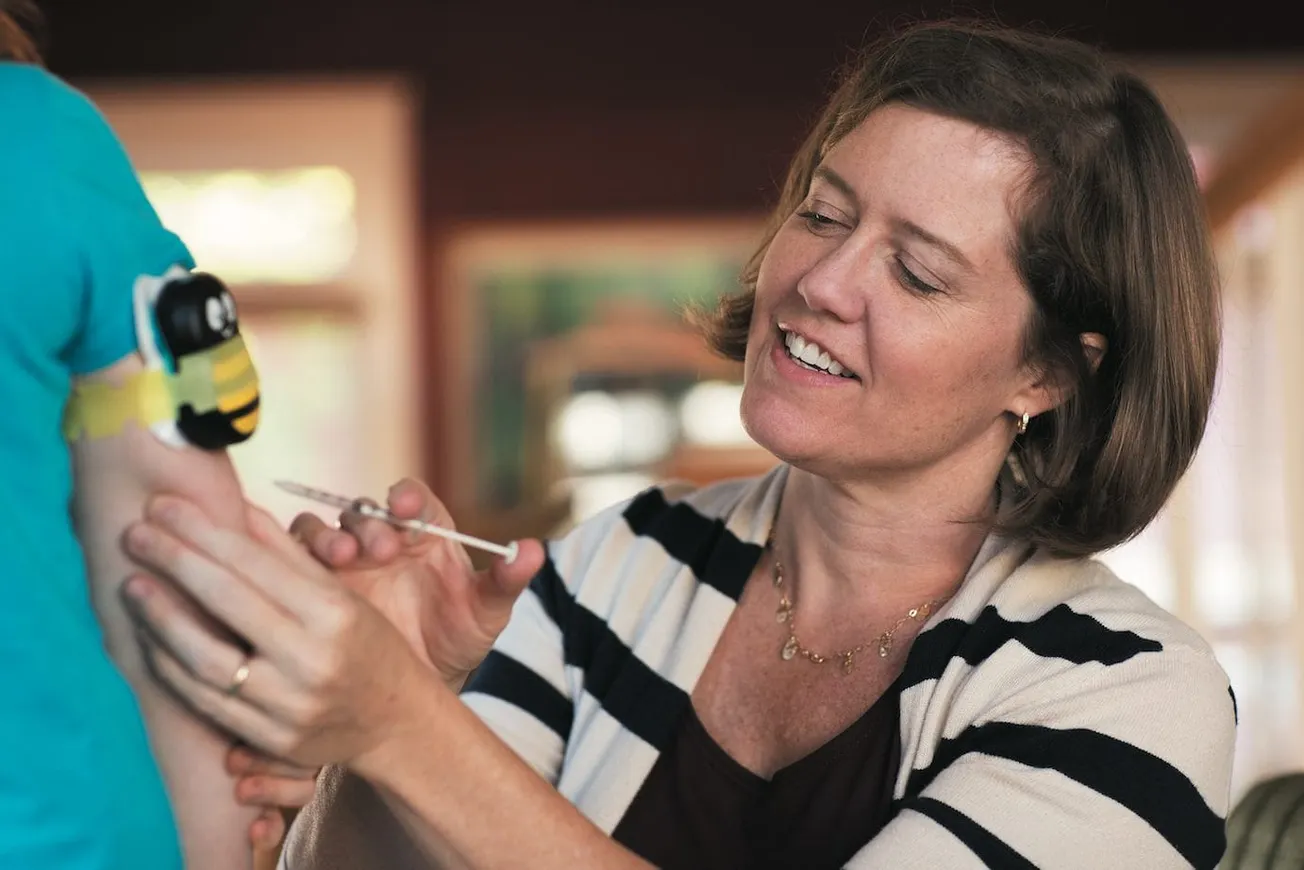
Needle fear contributed to an estimated 12,000 influenza deaths in the 2017-2018 flu season and impacts biologic adherence for 86% of patients. A new study out this month demonstrates a fast, inexpensive device called “Buzzy” that is widely used for IV access in hospitals may change that.

Walgreens and LabCorp announced the introduction of LabCorp at Walgreens into the California Inland Empire, with nine new LabCorp patient service centers within Walgreens stores in the greater Riverside, Calif. area.

A new drug pricing rule, proposed by the Centers for Medicare and Medicaid Services (CMS) and released Monday, Nov.

The National Pharmaceutical Council (NPC) today announced that Richard Bagger, executive vice president, corporate affairs and market access, Celgene Corp., has been elected chair of NPC’s board of directors for 2018-2019.

WOONSOCKET, R.I. – CVS Health today announced that it has completed its acquisition of Aetna establishing CVS Health as the nation’s premier health innovation company.

From Thanksgiving Day through Cyber Monday, more than 165 million Americans shopped either in stores or online, surpassing the 164 million who had said they would shop in a consumer sentiment survey conducted ahead of the holiday, the National Retail Federation and Prosper Insights & Analytics said.

This holiday season, PepsiCo is offering limited-time snack and beverage products for consumers to share and enjoy with friends and family.
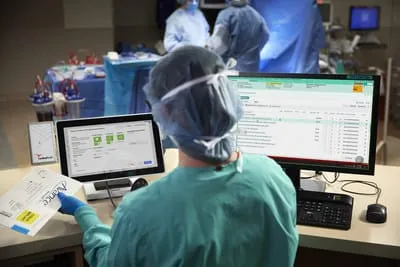
Cardinal Health inventory management solutions (also known as WaveMark) has introduced a supply chain automation solution designed for the unique challenges hospitals face in operating rooms (OR).

WASHINGTON – The National Community Pharmacists Association, the National Association of Chain Drug Stores, and the National Association of Specialty Pharmacy issued the following statement regarding the Centers for Medicare & Medicaid Services’ (CMS) release on Monday of the proposed rule titled “M

Teva Pharmaceutical Industries announced the release of limited doses of the FDA-approved generic version of EpiPen (epinephrine injection, USP) Auto-Injector, 0.3 mg, in the U.S. Teva’s generic version of the EpiPen Jr (epinephrine injection, USP) Auto-Injector, 0.

Diplomat Pharmacy is now shipping temperature-sensitive medication in ClimaCell coolers from TemperPack. ClimaCell coolers are fully recyclable at curbside and insulated with renewable materials that can be recycled wherever plastic bags are accepted.

At the upcoming 2018 American Society of Health System Pharmacists (ASHP) Midyear Clinical Meeting (December 2-6), Cardinal Health will highlight a comprehensive approach that addresses the top concern of hospital and health system pharmacy leaders: sustainable cost control.