
ProBioGen collaborates with Boehringer Ingelheim
ProBioGen announced on Tuesday a new non-exclusive collaboration with Boehringer Ingelheim on ProBioGen’s DirectedLuck transposase technology.

ProBioGen announced on Tuesday a new non-exclusive collaboration with Boehringer Ingelheim on ProBioGen’s DirectedLuck transposase technology.

As we look toward 2023 and a sense of normalcy that has eluded us for the past few years, there are several big things on the horizon to keep an eye on in pharmacy, medication use and high-quality, value-based care.

The U.S. Food and Drug Administration approved Leqembi (lecanemab-irmb) via the accelerated approval pathway for the treatment of Alzheimer’s disease. Leqembi is the second of a new category of medications approved for Alzheimer’s disease that target the fundamental pathophysiology of the disease.
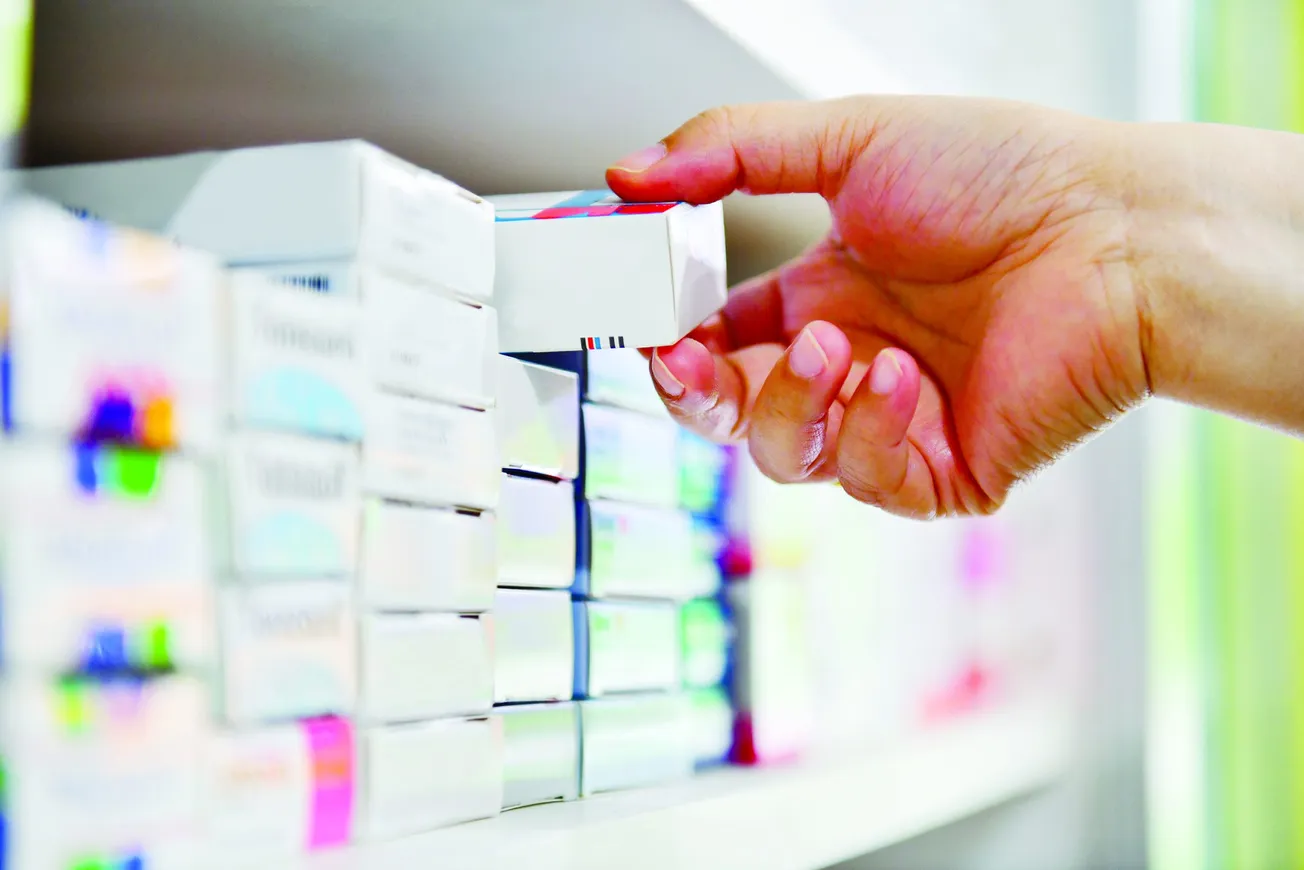
Specialty pharmacy continues to be dynamic, demanding and disruptive, undergoing rapid and dramatic change.

Walgreens’ overarching strategy is built on a combination of health and wellness expertise and exceptional convenience, says senior vice president and chief merchandising officer Luke Rauch.

PITTSBURGH – Science-backed ingredients and powerful flavors are synonymous with GNC Total Lean. The brand – which includes products across categories including meal replacements, weight management supplements and savory snacks – is built with science-backed, results-driven formulations.

Last year I reported that the COVID-19 pandemic drove tremendous growth in demand for over-the-counter medicines for pain and fever, dietary supplements for immune support and wellness, and consumer medical devices for self-diagnosis and protection.

In recent decades, pharmacy benefit managers have grown into shadowy and hugely influential players in the prescription drug supply chain. For the most part, they have managed to avoid public scrutiny and government regulation.

EAST WINDSOR, N.J. – Aurobindo Pharma Limited has received final approval from U.S. Food and Drug Administration for its Abbreviated New Drug Application lacosamide tablets USP, 50mg, 100mg, 150mg, and 200mg.

As the COVID-19 pandemic enters its third year, the public health emergency (PHE) declaration that has underpinned the country’s COVID-19 response efforts by providing the federal government with emergency powers, regulatory flexibilities and additional resources is currently set to expire in early

This week’s Video Forum features two leaders of the newly minted Haleon.

It is easy for those who are not health care professionals to overlook the tremendous potential for innovation in our field today. Similarly, anyone who is not a health policy wonk might not see the important ways policy is evolving to improve the delivery of care for millions of patients.

The pharmacy profession and patient care services delivered by the pharmacy team have evolved significantly over the past few years, bringing new opportunities and challenges.

EAST WINDSOR, N.J. – Aurobindo Pharma Limited has received final approval from U.S. Food and Drug Administration for its Abbreviated New Drug Application Dimethyl Fumarate Delayed-Release Capsules, 120mg and 240mg.

A contentious midterm election is behind us, and while pundits and other experts argue about what the results mean, those of us who deeply care about sound health care policy can hope for a new year where solid evidence and the quest for transparent information matters at least as much as political

Bausch + Lomb Corp. announced the U.S. introduction of PreserVision AREDS 2 Formula mini soft gels with OCUSorb. This product is the only AREDS 2 eye vitamin with the OCUSorb formulation.