
Pharmacy groups advocate USP standards for biologics
A coalition of pharmacy industry organizations has asked congressional leaders to weigh proposals that would free certain biologic drugs from adherence to U.S. Pharmacopeia (USP) standards.

A coalition of pharmacy industry organizations has asked congressional leaders to weigh proposals that would free certain biologic drugs from adherence to U.S. Pharmacopeia (USP) standards.

Pharmacy automation provider Innovation has entered into a technology integration agreement with IMA North America.

Mylan N.V. this week introduced two new generic drugs in the U.S. market. On Monday, the company announced final Food and Drug Administration approval for dextroamphetamine sulfate extended-release capsules — in dosages of 5 mg, 10 mg and 15 mg — and finasteride tablets 1 mg.

Steven Dinh has joined CutisPharma as executive vice president and chief scientific officer. CutisPharma said Monday that Dinh will play a key role at the company, overseeing its efforts to develop its pipeline of drugs for New Drug Application (NDA) submission to the Food and Drug Administration.
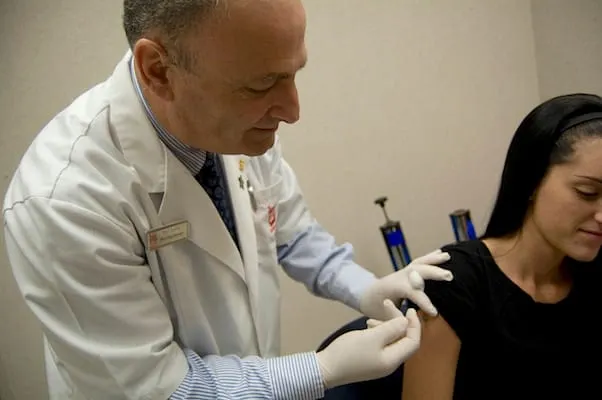
Walgreens has begun providing bacterial meningitis vaccinations at all of its pharmacies and in-store clinics on a walk-in basis. The drug chain said Monday that the new meningitis B vaccines, Bexsero and Trumenba, protect against most strains of serogroup B meningococcal disease.

Over the weekend AmerisourceBergen Corp. wrapped up its annual ThoughtSpot trade show, where it introduced new programs to help members of its Good Neighbor Pharmacy network drive their businesses.

Sanofi has gained Food and Drug Administration approval for Adlyxin (lixisenatide), a medication for type 2 diabetes.

CVS Health has partnered with Adapt Pharma to make the opioid overdose-reversal nasal spray Narcan more affordable for uninsured patients at CVS Pharmacy stores.
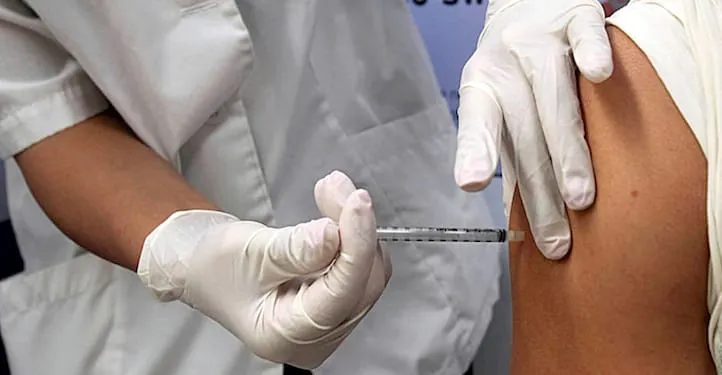
Bartell Drugs, Quality Food Centers (QFC), Hy-Vee and Giant Eagle are participating in National Association of Chain Drug Stores projects to demonstrate value-based payment models for pharmacy-administered immunizations.
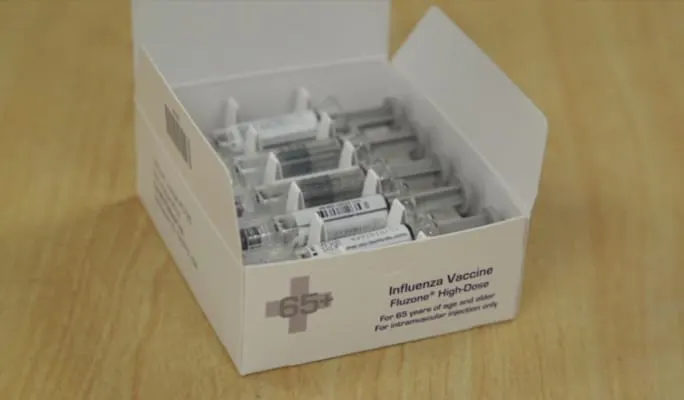
Sanofi Pasteur is shipping its first doses of Fluzone vaccines for the 2016-17 flu season following their release by the Food and Drug Administration. Sanofi’s vaccines division said the shipment marks the start of more than 65 million doses of seasonal flu shots that it will deliver to U.S.
The Federal Trade Commission has given its approval to two major pharma acquisitions: Teva Pharmaceutical Industries Ltd.’s $40 billion deal to buy Allergan plc’s generics business and Mylan N.V.’s $9.9 billion deal to acquire Meda AB.

Glenmark Pharmaceuticals Inc. USA has received final Food and Drug Administration approval for potassium chloride extended-release tablets.

Promius Pharma LLC, a subsidiary of Dr. Reddy’s Laboratories, has unveiled its new visual identity and corporate brand: The Power of Humans Being.

House Speaker Paul Ryan (R., Wis.) visited a Walgreens store in Wisconsin to mark the drug chain’s first store in the state with a drug take-back kiosk. Ryan attended an event on Monday at Walgreens’ store in Janesville, Wis.

Sandoz has launched rosuvastatin calcium tablets, a cholesterol-lowering medication, in the United States.

A group of congressmen have called on House of Representatives and Senate leaders to advance a TRICARE pilot that would enable military veterans and families to opt to fill prescriptions at community pharmacies.