
Cardinal Health to buy Harvard Drug Group
Cardinal Health Inc. plans to acquire pharmaceutical distributor The Harvard Drug Group (THDG) from private equity firm Court Square Capital Partners for $1.115 billion.

Cardinal Health Inc. plans to acquire pharmaceutical distributor The Harvard Drug Group (THDG) from private equity firm Court Square Capital Partners for $1.115 billion.

This month, Fruth Pharmacy plans to help demonstrate a new talking prescription label for the visually impaired. The regional drug chain has rolled out the AccessaMed prescription label to all of its 26 stores in West Virginia and Ohio.
Mylan N.V. and Teva Pharmaceutical Industries Ltd. have released guanfacine extended-release tablets, a nonstimulant medication for attention deficit hyperactivity disorder (ADHD), in the United States.

Lannett Co. has completed its acquisition of Silarx Pharmaceuticals Inc, , a manufacturer and marketer of liquid generic drug products. The deal also includes a related real estate entity, Lannett said Tuesday.
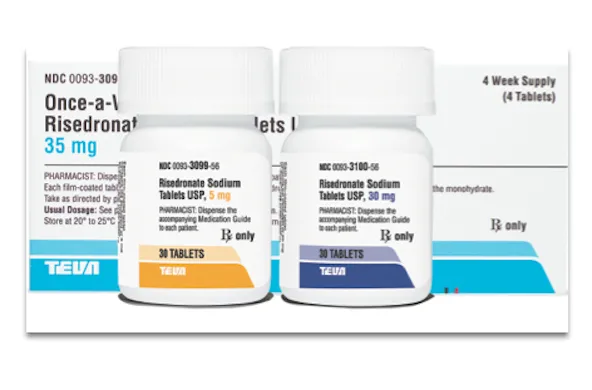
Teva Pharmaceutical Industries Ltd. has released generic versions of Actavis’ Actonel tablets, an osteoporosis medication, and of Amneal’s Lomedia 24 Fe, a women’s contraceptive, in the United States.

AmerisourceBergen Corp. is set to begin construction on three new distribution centers, to be located in Olive Branch, Miss.; Shakopee, Minn.; and Newburgh, N.Y. AmerisourceBergen said Monday that building for the new facilities is slated to start this summer and take 18 to 24 months to complete.

RxMedic Systems has introduced a new pharmacy robot, the RM200, which is engineered to use exclusive TriMaxx triangular vials. The pharmacy automation provider, a subsidiary of JM Smith Corp.

Walgreens Infusion Services is now Option Care, an independent company and brand, though Walgreens has retained “significant” minority ownership. Madison Dearborn Partners (MDP), a private equity firm based in Chicago, now owns the lion’s share of the new infusion services provider.
The Generic Pharmaceutical Association (GPhA) and its recently formed Biosimilars Council have praised the enactment of new legislation in five states to allow automatic substitution for Food and Drug Administration-approved interchangeable biologic products.

Eli Lilly and Co. has been cleared by the Food and Drug Administration to market the Humalog 200 units/ml KwikPen, which the company called the first concentrated mealtime insulin analog approved in the United States.

Boehringer Ingelheim has received approval from the Food and Drug Administration for Stiolto Respimat Inhalation Spray, a medication for chronic obstructive pulmonary disease (COPD).

A law that makes Washington the first state to grant provider status to pharmacists goes into effect this summer. Washington Gov. Jay Inslee (D.

CVS Health is entering a new market segment with a $12.7 billion deal to acquire Omnicare, a provider of pharmacy services to long-term care facilities.

Meijer Inc. has entered an agreement to buy Aureus Health Services, a national specialty pharmacy and health management service provider. Financial terms of the deal weren’t disclosed.

Walgreens and the National Urban League have kicked off their annual Wellness Tour, which brings free preventive and early-detection health services to urban and at-risk communities nationwide.

Mike Podgurski, vice president of pharmacy services at Rite Aid Corp., has been named the 2015 Honorary President by the National Association of the Boards of Pharmacy (NABP).