
Support public school teachers with BIC this back to school season
Back to school season is here and parents are flooding stores to stock up on school supplies for a successful year ahead.

Back to school season is here and parents are flooding stores to stock up on school supplies for a successful year ahead.

NEW YORK– Bud Light and the Cleveland Browns announced the creation of the Bud Light Cleveland Browns “Victory Fridge,” a smart-technology refrigerator that will automatically unlock after the Browns capture their first regular-season victory of the 2018 NFL season.

ATLANTA and WHITESTONE, N.Y. – The Coca-Cola Co. and BodyArmor announced that they have entered into a definitive agreement through which Coca-Cola will acquire a minority ownership stake in BodyArmor.

Ranked by SPINS as the United States’ No. 1 fastest growing natural toothbrush brand (now in over 15,000 stores worldwide), WooBamboo’s “eco-awesome” oral care products were designed to inspire the world and empower eco-minded consumers.

Today S.Pellegrino Sparkling Natural Mineral Water, recognized as a leader of iconic taste and authentic Italian style, introduces sleek cans to their product lineup. Delivering the same refreshing mix of gentle bubbles and fine mineral balance customers know and love, the new S.

Mylan N.V. announced the U.S. launch of tadalafil tablets USP, 20 mg, the first generic version of the reference listed drug, Eli Lilly and company’s Adcirca. Mylan Pharmaceuticals received final approval from the U.S.

HAUPPAGUE, N.Y.– JD Beauty Group announces the acquisition of Ouidad, the curl expert hair care and styling brand. Popular among North American salon professionals, the company will join the JD Beauty group of industry-leading hair brands WetBrush and Bio Ionic.

Today’s big sexy world is confusing and challenging, resulting in endless questions and making intimate conversations around sex more complicated than ever.
Kellogg Co. is providing cereals, bars and snacks to California families forced to evacuate their homes due to wildfires that continue to rage. Five semi-trailers, or more than 1.7 million servings, of Kellogg foods are being delivered to area food banks by its charity partner, Feeding America.

Parents want to feel good about what they put on their baby’s skin — and so does Johnson’s Baby. That’s why the brand is disclosing 100% of the ingredients in its found in its baby products—including those found in fragrances.

Football fans all over the country can rejoice: Bud Light’s NFL team cans are back just in time for kick-off. As the official beer of the NFL, Bud Light is introducing new designs for cans that feature a team logo, as well as a customized color stripe down the side, enabling fans

Apothecary Products, a leading innovator, manufacturer and distributor of consumer health care products, has announced the acquisition of SOFT Style, based in Boulder, Colo. SOFT Style develops, manufactures and markets a wide range of breastfeeding support products under the bamboobies brand.
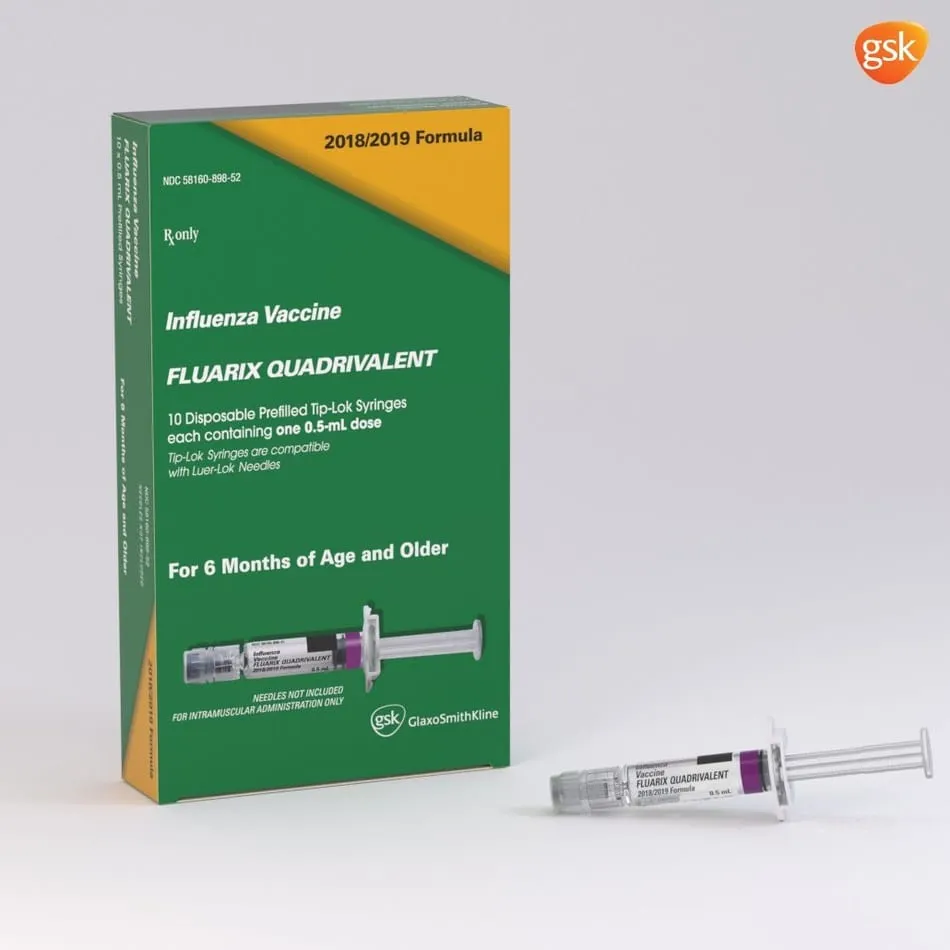
GlaxoSmithKline (GSK) today announced it will begin shipping its quadrivalent influenza vaccines to US healthcare providers and pharmacies for the 2018-19 flu season, immediately following licensing and lot-release approval from the US Food and Drug Administration’s (FDA) Center for Biologics Evalua

Following the spectacular success of Finishing Touch Flawless, the 2018 Women’s Grooming Product of the Year and the No.1 best-selling women’s facial hair removal device, the Brand announced today the launch of its first brow maintenance tool, Flawless Brows.
GlaxoSmithKline (GSK) announced that Iain Mackay has been appointed GSK’s next chief financial officer (CFO). He has also been appointed as an Executive Director to the GSK board. Mackay will join the company on January 14, 2019.

Rice Krispies Treats has partnered with the National Federation of the Blind to create the first-ever accessible ‘Love Notes’ in the form of Braille stickers and re-recordable audio boxes so parents and family members can share messages of love and encouragement with children who are blind or low-vi