
Pharmacy


NACDS gives input on HHS’ Healthy People 2030 initiative
The National Association of Chain Drug Stores (NACDS) has submitted comments containing recommendations for the Department of Health and Human Services’ (HHS) Healthy People 2030 initiative, which develops a new set of evidence-based, 10-year national objectives aimed at improving the health of all

Upsher-Smith celebrates 100 years of serving patients
MAPLE GROVE, Minn. – Upsher-Smith proudly kicked off its centennial anniversary this month at its headquarters here.

ACPE elects new officers, announces new board members
CHICAGO – The Accreditation Council for Pharmacy Education (ACPE) announced that Lori Duke, Anthony Provenzano, and Michael Moné have been elected as members of the ACPE executive committee for the 2019–2020 term of office.

Dr. Reddy’s gets FDA approval for Tosymra
Dr. Reddy’s Laboratories and its subsidiary, Promius Pharma. today announced the approval of Tosymra (previously known as DFN-02) by the U.S. Food and Drug Administration (FDA). Tosymra is indicated for the acute treatment of migraine with or without aura in adults.

Walgreens gets the largest share of Shopko auction assets
NEW YORK – Walgreens Boots Alliance was the big winner in the auction for the pharmacy assets of bankrupt retailer Shopko, taking charge of about half the assets that were on sale.

Jhaveri named Health Mart president
Nimesh Jhaveri has joined the Health Mart family of community pharmacies as president. Jhaveri, who has 30 years of hands-on pharmacy experience, started in his new role on October 31.

NACDS demonstrates need for DIR fee reform in comments to CMS
Based on insights from member pharmacies that serve as the front lines of healthcare delivery, the National Association of Chain Drug Stores (NACDS) has submitted extensive and thoroughly documented comments to the Centers for Medicare & Medicaid Services in support of the agency’s proposal to refor
The profession of pharmacy: A tale of two realities
Editor’s note: This was part of CDR’s 2019 Pharmacy Outlook in the January 7 issue. Today, pharmacists are facing a health care environment that presents both opportunities and challenges.

CVS Health announces transaction with Vitality Re X
CVS Health announced that Aetna, a CVS Health business, has entered into a four-year reinsurance arrangement with Vitality Re X Limited as part of its long-term capital management strategy.

Hy-Vee dietitians to provide free biometric screenings in February
For the second year, Hy-Vee, Inc. will be providing free biometric screenings throughout its eight-state region during Heart Health Month in February. Hy-Vee dietitians working out of Hy-Vee Healthy You Mobiles will offer the free biometric screenings to customers at 150 Hy-Vee stores.

CVS announces organizational changes
CVS Pharmacy has expanded the responsibilities of Judy Sansone, the chain’s senior vice president of front store business and chief merchant. In addition, George Coleman has been promoted to senior vice president of merchandising, reporting to Sansone.
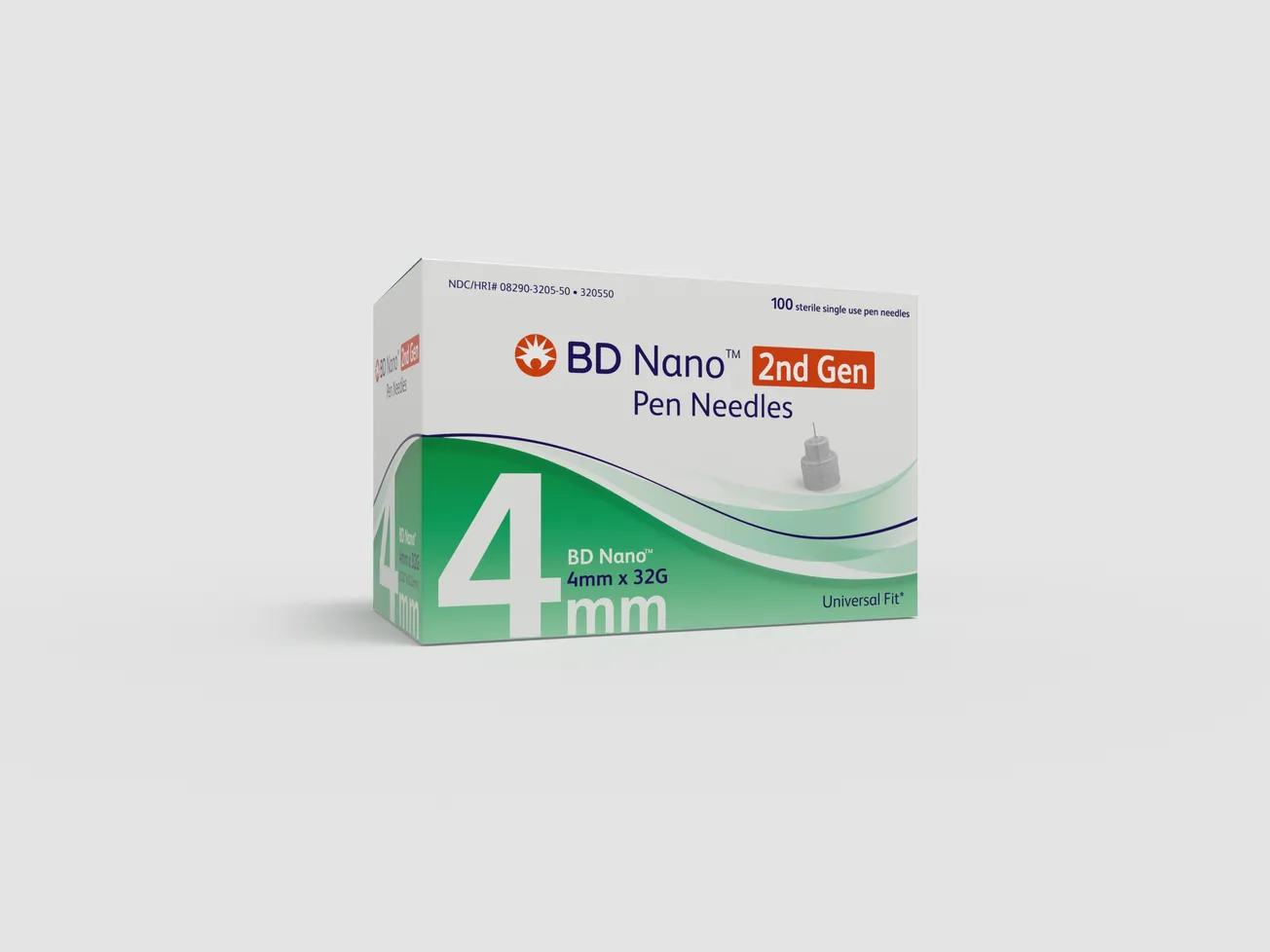
BD launches ergonomic pen needle technology
BD (Becton, Dickinson and Company), announced the U.S. Food and Drug Administration 510(k) clearance of its second generation BD Nano pen needle, designed for more reliable subcutaneous injection depth.

Canada’s pharmacy sector holding strong
Editor’s note: This was part of CDR’s 2019 Pharmacy Outlook in the January 7 issue. Looking ahead to 2019, the pharmacy business is poised to be more disrupted than ever before.

Aurobindo receives FDA approval for dofetilide capsules
Aurobindo Pharma has received final approval from U.S. Food and Drug Administration for its Abbreviated New Drug Application for Dofetilide Capsules, 0.125 mg, 0.25 mg, and 0.5 mg. Aurobindo’s dofetilide capsules are an AB-rated generic equivalent to the reference listed drug, Pfizer’s Tikosyn.

Hoy Health launches first international prescription gift vouchers
MORRISTOWN, N.J. – For the first time, Hoy Health makes it possible for consumers to purchase medications in the U.S for their loved ones in Latin America, and Hoy Health will home-deliver within 24 hours. Consumers can visit HoyRX.
