
Teva launches of a generic version of Uceris in the United States
Teva Pharmaceutical Industries Ltd., today announced the launch of a generic version of Uceris (budesonide) extended-release tablets, 9 mg, in the U.S.

Teva Pharmaceutical Industries Ltd., today announced the launch of a generic version of Uceris (budesonide) extended-release tablets, 9 mg, in the U.S.

McKesson ideaShare 2018, McKesson Corporation’s annual interactive forum for community pharmacists, kicked off today in Las Vegas. The annual event offers those in community pharmacy the opportunity to improve their knowledge, performance and profitability while networking with industry experts.
Driven by the 21st Century Cures Act, Food and Drug Administration Commissioner Scott Gottlieb has publicly stated a goal of moving the FDA into the new century by creating a regulatory process that is modern and efficient.

BOISE, Idaho – Albertsons Cos. announced this week their Idaho pharmacists can now prescribe for more health conditions under a new state law. Prior to the law’s July 1 effective date, targeted Albertsons Cos.

Pharmaceutical Care Management Association (PCMA) president and chief executive officer Mark Merritt released the following statement on reports of yet another round of massive price hikes by drug manufacturers.


SOMERSET, N.J. – Catalent, a leading global provider of advanced delivery technologies and development solutions for drugs, biologics and consumer health products, announced that it has agreed to acquire Juniper Pharmaceuticals, including its Nottingham, U.K.-based Juniper Pharma Services division.

Amazon’s announced acquisition of PillPack gives the tech giant a vehicle for disrupting the U.S. drug distribution market – and may come with a beneficial side-effect for pharma. Amazon will find substantial reception for its Rx distribution services among U.S. patients and physicians.
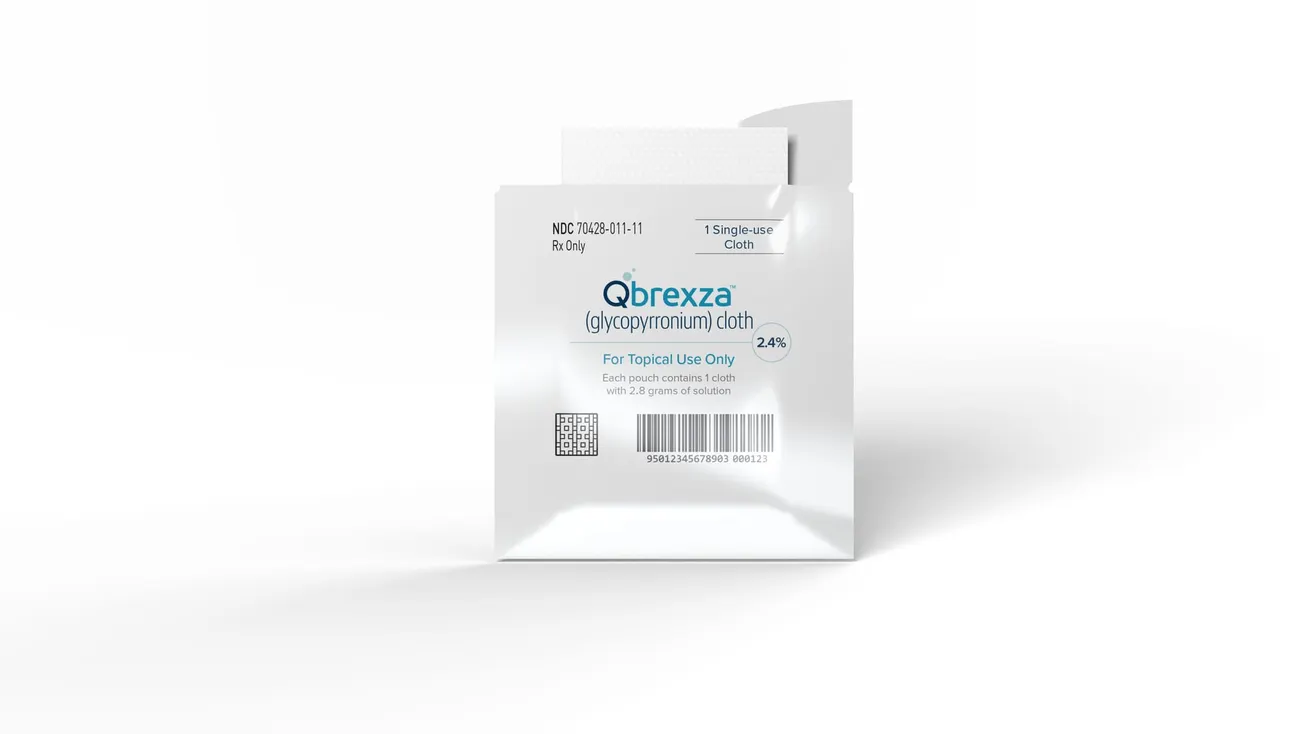
Dermira announced that the U.S. Food and Drug Administration (FDA) has approved Qbrexza (glycopyrronium) cloth, an anticholinergic indicated for the topical treatment of primary axillary hyperhidrosis in adult and pediatric patients 9 years of age and older.

Bartle’s Pharmacy of Oxford, N.Y., received McKesson’s Pharmacy of the Year Award. The Health Mart community pharmacy is being recognized for their superior clinical performance, high-touch patient relationships and proactive advocacy efforts.

Pharma major Lupin and Mylan announced that the two companies will partner to commercialize a biosimilar to Enbrel (etanercept).

Amazon and PillPack today announced that they have entered into a definitive merger agreement under which Amazon will acquire PillPack. PillPack is a pharmacy said to be designed to provide the best possible customer experience in the U.S. for people who take multiple daily prescriptions.

Avella has announced that it will distribute Braftovi (encorafenib and Mektovi (binimetinib), a combination therapy recently FDA approved for patients for the treatment of patients with unresectable or metastatic melanoma.

Aurobindo Pharma has launched atazanavir capsules 100 mg, 150 mg, 200 mg and 300 mg upon receiving final approval from the U.S. Food and Drug Administration for its Abbreviated New Drug Application.

The National Association of Chain Drug Stores (NACDS), the Washington State Pharmacy Association (WSPA) and the National Community Pharmacists Association (NCPA) filed a legal brief this week with the Washington State Court of Appeals, challenging the state’s refusal to reimburse community pharmacie

Albertsons Cos. announced that more than 1,600 of its pharmacies across 21 states have pharmacists with the ability to independently provide naloxone, a medication used to reverse the impacts of an opioid overdose, and to educate patients and caregivers about its use.