
Lupin and Mylan team up to commercialize Enbrel
Pharma major Lupin and Mylan announced that the two companies will partner to commercialize a biosimilar to Enbrel (etanercept).

Pharma major Lupin and Mylan announced that the two companies will partner to commercialize a biosimilar to Enbrel (etanercept).

Avella has announced that it will distribute Braftovi (encorafenib and Mektovi (binimetinib), a combination therapy recently FDA approved for patients for the treatment of patients with unresectable or metastatic melanoma.

In order to apply sunscreen to hard to reach areas such as the back, neck, shoulders, sides, hips, legs and feet, Starco Brands introduces Honu, the first and only sunscreen with a patented spray wand.

Aurobindo Pharma has launched atazanavir capsules 100 mg, 150 mg, 200 mg and 300 mg upon receiving final approval from the U.S. Food and Drug Administration for its Abbreviated New Drug Application.

Soleo Health, an innovative leader in specialty infusion services, announced the company has been selected by Genentech (member of the Roche Group), a world-leading biotechnology company with multiple products on the market and a strong development pipeline, to dispense two limited distribution drug

Kimberly-Clark Corp. has reported its progress toward achieving its Sustainability 2022 strategy to create social, environmental and business value.

HeadBlade, a leader in HeadCare and developers of the ultimate head shaving razor, the HeadBlade MOTO, announced Tom Wainman, formerly a pitchman for Hair Club for Men, as its new spokesperson.
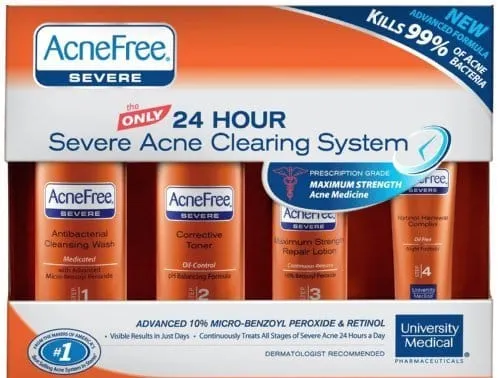
AcneFree announces its support of the American Acne and Rosacea Society (AARS), a non-profit organization, as a part of its commitment to better educate the 21 million teenagers in the U.S. – approximately 80% of all teens – who suffer from acne and occasional breakouts.

Upsher-Smith Laboratories (Upsher-Smith) announced that it will be featuring Platinum Pass, the latest addition to the Access Pathways Program for Qudexy XR (topiramate) extended-release capsules at the 60th Annual Scientific Meeting of the American Headache Society (AHS), June 28 to July 1, in

Genentech announced that the U.S. Food and Drug Administration (FDA) has accepted a New Drug Application (NDA) and granted Priority Review for baloxavir marboxil as a single-dose, oral treatment for acute, uncomplicated influenza in people 12 years and older.

Aurobindo Pharma announced that the company has received final approval from the U.S. Food & Drug Administration to manufacture and market ertapenem injection 1 g/vial. Aurobindo’s ertapenem injection is a generic equivalent of Merck Sharp & Dohme Corp’s Invanz Injection.

Starting next month, Dove will be taking another step in ensuring beauty is a source of confidence and not anxiety with the launch of a ‘No Digital Distortion Mark’. The Mark will roll out across all branded content globally beginning in July with deodorant campaigns leading the initiative.

Dove Chocolate is hosting the first-ever Cocoa Farmers Market at Santa Monica Place in Los Angeles, an interactive experience detailing the chocolate-making journey from bean to bar.

Earlier this year, Harmony Herbal introduced Skin Therapy Calm and Calm + Detox, said to be the first herbal medicine-based supplement and topical combination duo to relieve the symptoms of rash, eczema and dermatitis with a whole body wellness approach.

To celebrate the roll out of new Jif Power Ups peanut butter snacks, the brand is teaming with multi-talented award-winning actor and father of two, Neil Patrick Harris, to shed a humorous light on trending parenting challenges and introduce the ultimate win-win solution that is equal parts deliciou

Vizuri Health Sciences announced that the company has further strengthened its consumer healthcare operational and transactional experience by adding Elise Donahue to its board of directors.