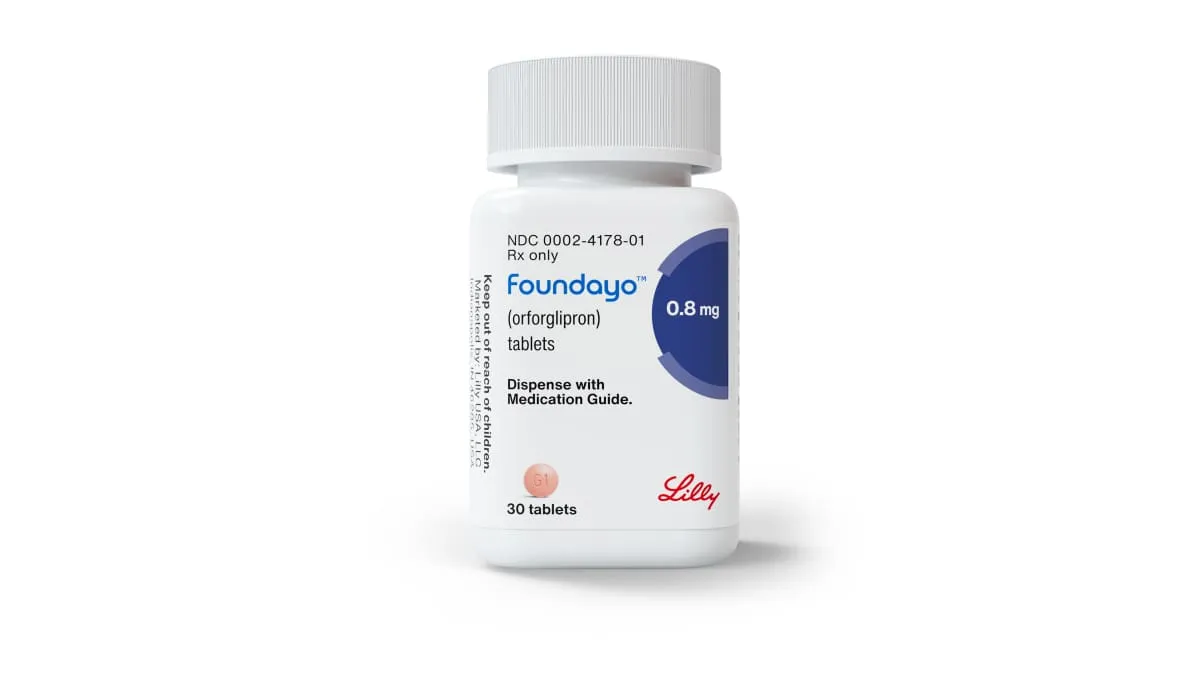
INDIANAPOLIS — Eli Lilly and Company announced that the U.S. Food and Drug Administration has approved Foundayo (orforglipron), a once-daily oral GLP-1 therapy for adults who are obese or overweight with related medical conditions. This marks the first weight-loss pill in its class that can be taken without food or water restrictions.
The approval positions Foundayo as a key addition to the rapidly expanding GLP-1 receptor agonist market, which has mainly been driven by injectable treatments. Lilly announced that the drug will be available through its direct-to-consumer platform LillyDirect with free home delivery starting April 6, with broader availability across U.S. retail pharmacies and telehealth providers to follow.
Clinical data from the ATTAIN program showed that patients taking the highest dose of Foundayo lost an average of 27.3 pounds, or 12.4% of their body weight, compared to 2.2 pounds for the placebo. Across the study population, participants achieved an average weight loss of about 25 pounds regardless of whether they completed the trial. The therapy also showed improvements in cardiovascular risk markers, including cholesterol levels, triglycerides, waist circumference, and systolic blood pressure.
“People living with obesity need treatment options that meet them where they are,” said Deborah Horn, DO, of UTHealth Houston, noting the flexibility of a once-daily oral option without dosing restrictions. Lilly CEO David A. Ricks added that the company aims to address barriers such as access, complexity and stigma that have limited broader adoption of GLP-1 therapies.
Lilly is positioning Foundayo as a more accessible entry point into obesity treatment. Eligible patients with commercial insurance may pay as little as $25 per month, while self-pay prices start at $149 monthly for lower doses. Medicare Part D coverage, costing around $50 per month, is expected to begin as early as July 2026.
The company warned that Foundayo should not be used with other GLP-1 therapies and issued a warning for potential thyroid tumors, including cancer. Common side effects include gastrointestinal issues such as nausea, diarrhea, and abdominal discomfort.
With submissions already filed in over 40 countries, Lilly plans to expand global availability after regulatory approvals, further increasing competition in the obesity treatment market as demand for GLP-1 therapies continues to rise.
Submit Your Press Release
Have news to share? Send us your press releases and announcements.
Send Press Release